
Note: Contraindications to dorsal column stimulation for intractable angina are presented in an Appendix to the Background section of this CPB. Reversible ischemia is documented by symptom-limited treadmill exercise test.Member’s angina pectoris is New York Heart Association (NYHA) Functional Class III (patients are comfortable at rest less than ordinary physical activity causes fatigue, palpitation, dyspnea, or anginal pain) or Class IV (symptoms of cardiac insufficiency or angina are present at rest symptoms are increased with physical activity) and.Optimal pharmacotherapy includes the maximal tolerated dosages of at least 2 of the following anti-anginal medications: long-acting nitrates, beta-adrenergic blockers, or calcium channel antagonists and 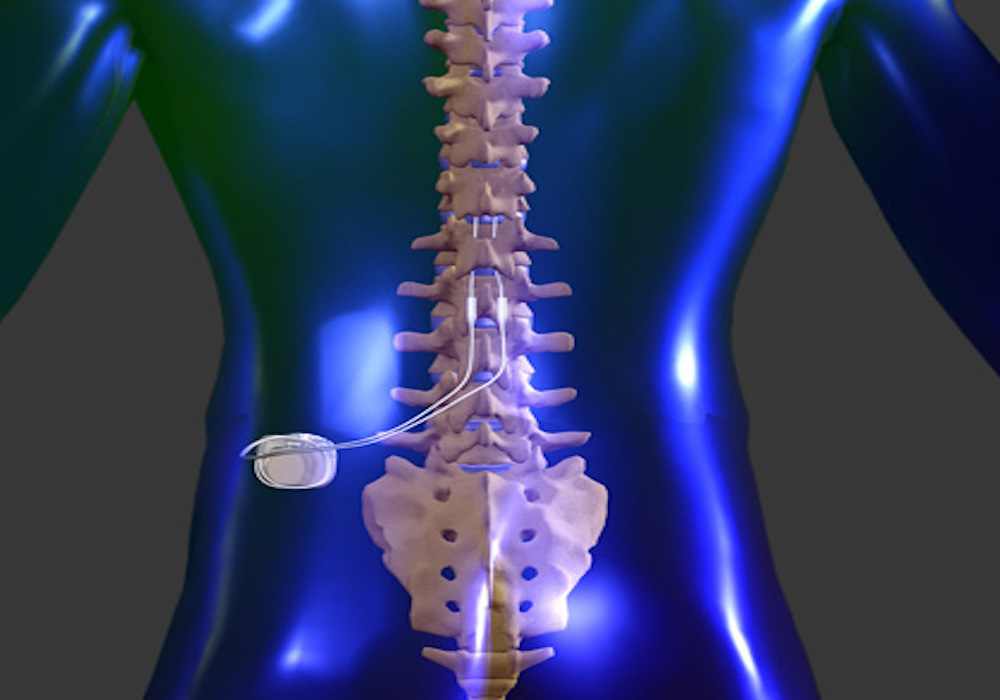
Member has had optimal pharmacotherapy for at least one month.Member has angiographically documented significant coronary artery disease and is not a suitable candidate for revascularization procedures such as coronary artery bypass grafting (CABG) or percutaneous transluminal coronary angioplasty (PTCA) and.(A trial of percutaneous spinal stimulation is considered medically necessary for members who meet the above-listed criteria, in order to predict whether a dorsal column stimulator will induce significant pain relief) and Member experienced significant pain reduction (50 % or more) with a 3- to 7-day trial of percutaneous spinal stimulation.DCS as DME for the management of intractable angina in members who are not surgical candidates and whose pain is unresponsive to all standard therapies when all of the following criteria are met:.The use of cervical dorsal column stimulation for the treatment of members with complex regional pain syndrome when criteria in section A are met and the member has experienced significant pain reduction (50 % or more) with a 3- to 7-day trial of percutaneous spinal stimulation.Implantation of a dorsal column stimulator (DCS) for members who meet the above-listed criteria who have experienced significant pain reduction (50 % or more) with a 3- to 7-day trial of percutaneous spinal stimulation.Member’s functional disability assessed using the Oswestry Disability Index (ODI) member has received an ODI score greater than or equal to 21%. 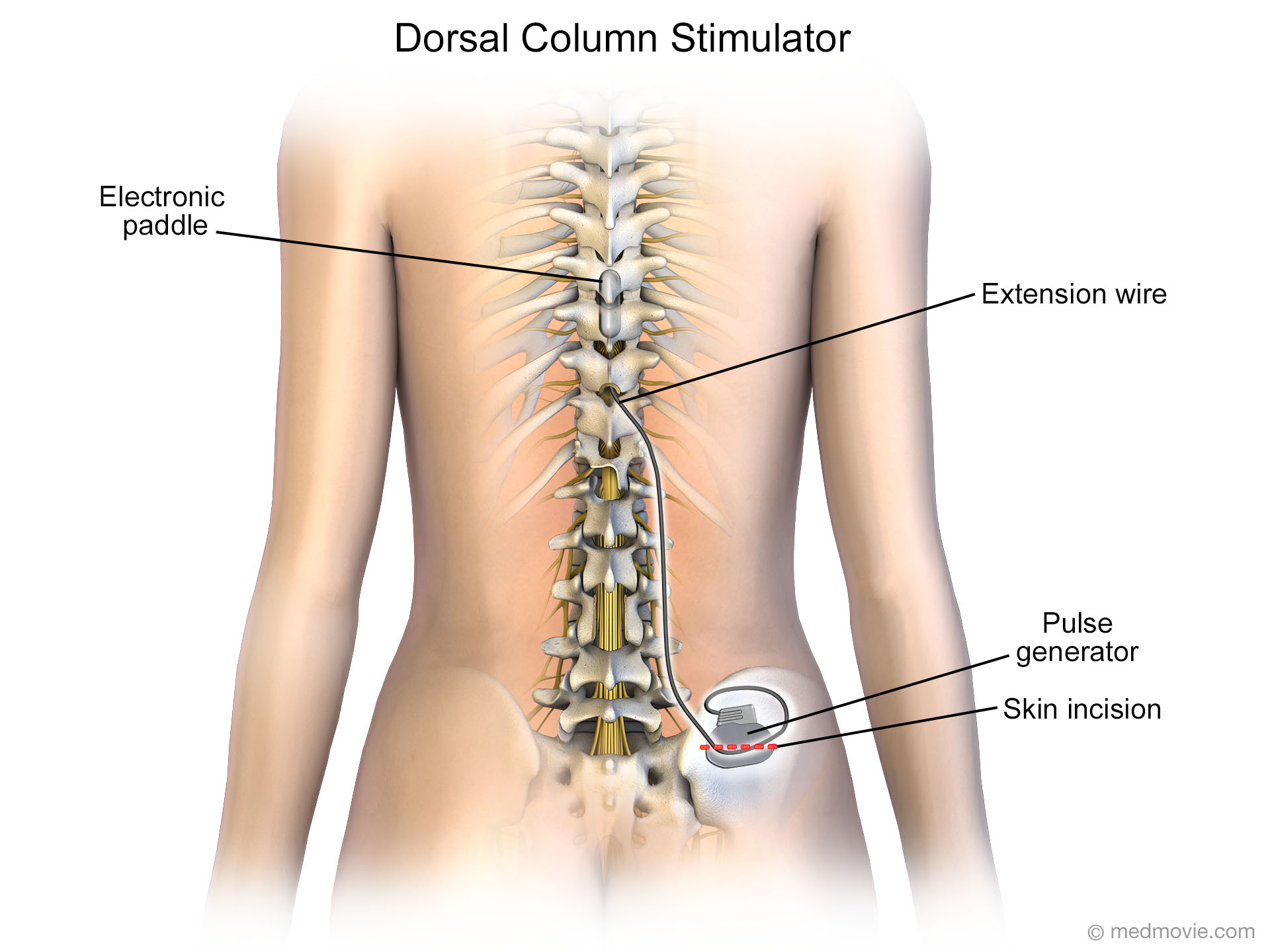
There is documented pathology, i.e., an objective basis for the pain complaint and.Other more conservative methods of pain management (including non-steroidal anti-inflammatory drugs, tricyclic antidepressants, and anticonvulsants) have been tried and failed for a minimum of 6 months and.Member has obtained clearance from a psychiatrist, psychologist, or other qualified mental health professional (e.g., Master of Social Work in behavioral health) and.Member does not have any untreated existing substance use disorder(s) (per American Society of Addiction Medicine (ASAM) guidelines), and.Member has undergone careful screening, evaluation and diagnosis by a multidisciplinary team prior to implantation (Note: screening must include psychological as well as physical evaluations) and.The member must meet all of the following criteria:.Last resort treatment of moderate to severe (5 or more on a 10-point VAS scale) chronic neuropathic pain of certain origins (i.e., lumbosacral arachnoiditis, phantom limb/stump pain, peripheral neuropathy (including diabetic peripheral neuropathy), post-herpetic neuralgia, intercostal neuralgia, cauda equina injury, incomplete spinal cord injury, or plexopathy) that has been present for 12 or more months and. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
